Organ-izing the cells
Team Leader : Vincent Studer

Vincent Studer is a CNRS research director at the Interdisciplinary Institute for Neuroscience (IINS) in Bordeaux. After completing an engineering degree at the ESPCI Paristech in 2001 he carried out his Ph.D at the Pierre & Marie Curie University in Paris in Material Science. His early research work on single cell microfluidics was performed both in the laboratory for Photonics and Nanostructure (LPN-CNRS) in Marcoussis and in the lab of Steve Quake at Caltech. He joined the CNRS in 2004 as a tenured scientist, first at the ESPCI Neurobiology lab and later (2009) at the IINS in Bordeaux. He is an expert in microengineering and microscopy for biology. He has authored more than 30 articles and has filed over 10 patents. He is also a co-founder of Alvéole, a company commercializing a biomolecule printer invented in his lab. He was awarded the CNRS bronze medal in 2013 for early career achievements and the Jerphagnon prize in 2017 for his successful tech transfer to Alvéole.
General objective
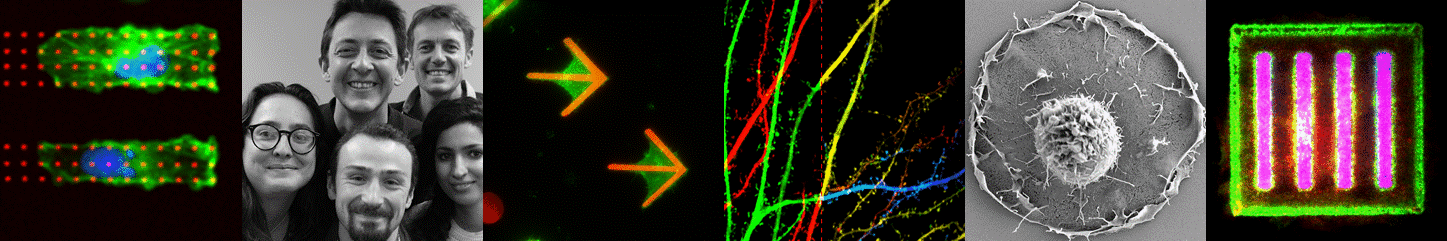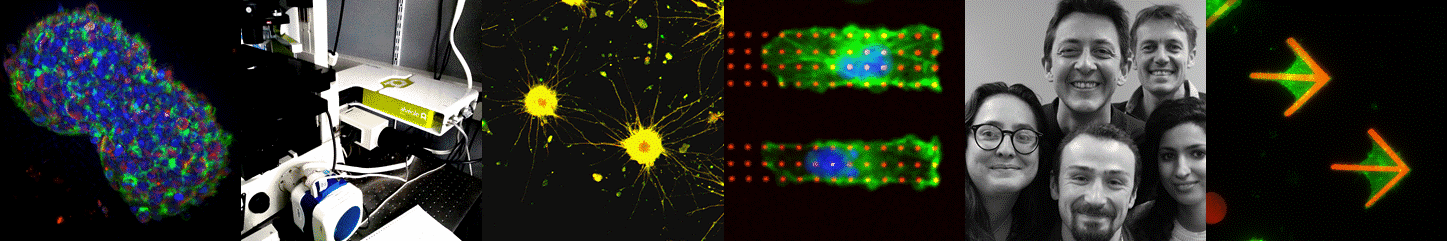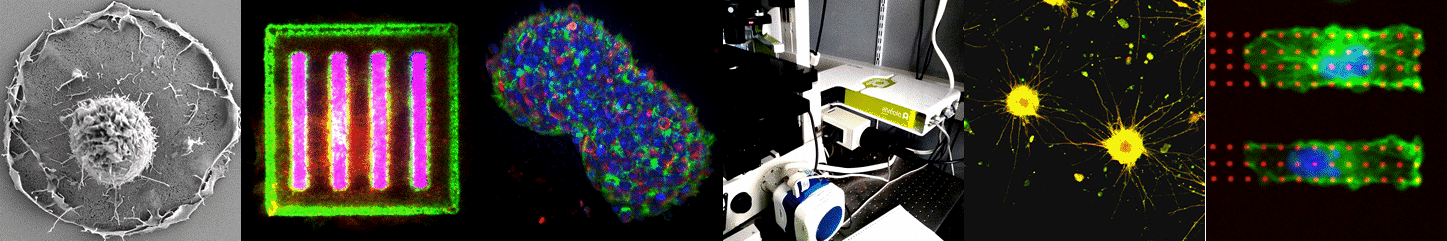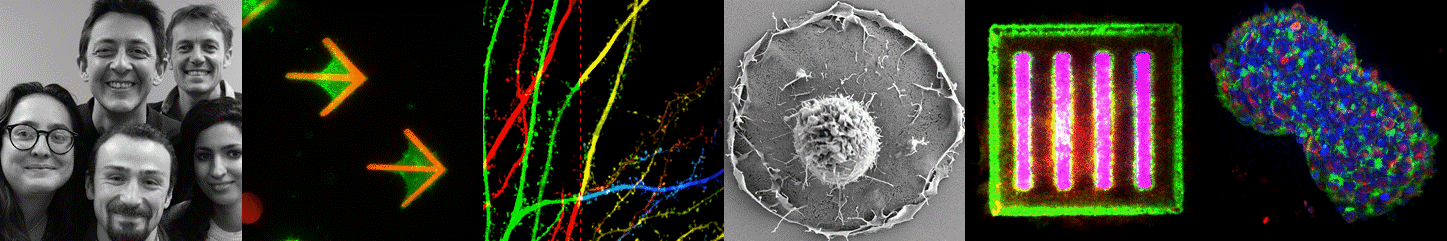
3D cell culture i.e. the art of organ-izing cells by cultivating them in configurations that more closely mimic the in-vivo environment is widely considered as the main route towards more physiologically relevant in vitro models. Yet providing simple, large scale and standardized solutions remains a key challenge for the widespread adoption of such cell-based models in laboratories and later in the pharmaceutical industry. Pioneers in the field of 3D brain cultures undoubtedly demonstrated the huge potential of such models in neuroscience. They also point out that novel tools and materials are required to advance their maturation (complexity) and scalability. This field of research is growing rapidly, mainly fueled by the establishing human Induced Pluripotent Stem cell technology. By joining our means and efforts in a joint research laboratory with the company Alvéole, we have established a tight research collaboration and an experienced innovation structure in the field of in vitro model design and experimentation. Together we aim at applying our patented hydrogel structuration and protein patterning toolbox to develop standardized in vitro models of human brain tissues. Such in vitro models, especially when their size is kept at the microscale, have the capacity to be observed and quantified by state of the art high resolution fluorescence microscopy and electron microscopy. A second aim of our research is to develop specific tools and methods to enable high troughput imaging of such human microtissues. This novel and exciting avenue to dissect fundamental mechanisms of human brain physiology in vitro will first be applied to study the dynamic of synaptic circuitry in collaboration with the other teams from IINS.
Expertise
Selected Publications
MORE
MORE
Members
« Researcher »
| CHAMMA Ingrid | Researcher | ingrid.chamma@u-bordeaux.fr | +33533514746 |  |
| STUDER Vincent | Researcher | vincent.studer@u-bordeaux.fr | +33533514744 |  |
« Technical Staff »
| DIAZ Candice | Technical staff | candice.diaz@u-bordeaux.fr | +33533514700 | |
| JEANNIERE Caroline | Technical staff | caroline.jeanniere@u-bordeaux.fr | +33533514700 |  |
« PhD student »
| CRESP Oscar | PhD student | oscar.cresp@u-bordeaux.fr | +33533514700 |